Summary of Biochemical Tests
Tests used to identify Gram Positive Bacteria
Tests used to identify Gram Negative Bacteria
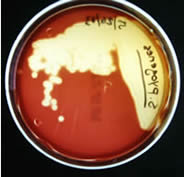
Mannitol Salt Agar (MSA)
This type of medium is both selective and differential. The MSA will select for organisms such as Staphylococcus species which can live in areas of high salt concentration (plate on the left in the picture below). This is in contrast to
Streptococcus species, whose growth is selected against by this
high salt agar (plate on the right in the picture below).
The differential ingredient in MSA is the sugar mannitol. Organisms
capable of using mannitol as a food source will produce acidic byproducts
of fermentation that will lower the pH of the media. The acidity of
the media will cause the pH indicator, phenol red, to turn yellow. Staphylococcus
aureus is capable of fermenting mannitol (left side of left plate)
while Staphylococcus epidermidis is not (right side of left plate).

TOP
Glucose
broth with Durham tubes
This is a differential medium. It tests an organism's ability
to ferment the sugar glucose as well as its ability to convert
the end product of glycolysis, pyruvic acid into gaseous byproducts.
This is a test commonly used when trying to identify Gram-negative
enteric bacteria, all of which are glucose fermenters but only
some of which produce gas.
Like MSA, this medium also contains the pH indicator, phenol red.
If an organism is capable of fermenting the sugar glucose, then
acidic byproducts are formed and the pH indicator turns yellow. Escherichia coli is capable of fermenting glucose as are Proteus mirabilis (far right) and Shigella dysenteriae (far left). Pseudomonas aeruginosa (center)
is a nonfermenter.
The end product of glycolysis is pyruvate. Organisms that are
capable of converting pyruvate to formic acid and formic acid
to H2 (g) and CO2 (g), via the action of the enzyme formic hydrogen lyase, emit
gas. This gas is trapped in the Durham tube and appears as a bubble
at the top of the tube. Escherichia coli and Proteus
mirabilis (far right) are both gas producers. Notice that Shigella dysenteriae (far left) ferments glucose but does not produce gas.
*Note - broth tubes can be made containing sugars other than
glucose (e.g. lactose and mannitol). Because the same pH
indicator (phenol red) is also used in these fermentation tubes,
the same results are considered positive (e.g. a lactose
broth tube that turns yellow after incubation has been inoculated
with an organism that can ferment lactose).
  
TOP
Blood Agar Plates (BAP)
This is a differential medium. It is a rich, complex medium that contains
5% sheep red blood cells. BAP tests the ability of an organism to produce
hemolysins, enzymes that damage/lyse red blood cells (erythrocytes).
The degree of hemolysis by these hemolysins is helpful in differentiating
members of the genera Staphylococcus, Streptococcus and
Enterococcus.
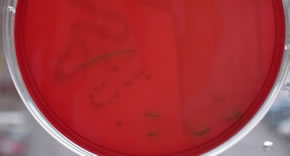
- Beta-hemolysis is complete hemolysis. It is characterized by a clear
(transparent) zone surrounding the colonies. Staphylococcus aureus, Streptococcus pyogenes and Streptococcus agalactiae are b-hemolytic (the picture on the left below
shows the beta-hemolysis of S. pyogenes).
- Partial hemolysis is termed alpha-hemolysis. Colonies typically are
surrounded by a green, opaque zone. Streptococcus pneumoniae and Streptococcus mitis are a-hemolytic
(the picture on the right below shows the a-hemolysis of S. mitis).
- If no hemolysis occurs, this is termed gamma-hemolysis. There are no
notable zones around the colonies. Staphylococcus epidermidis is gamma-hemolytic.
What type of hemolysis is seen on each one of the following
plates?
 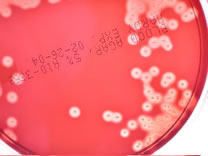 
TOP
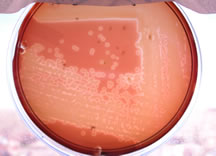
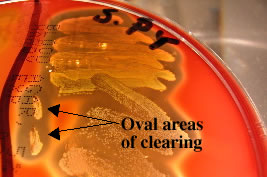
Streak-stab
technique
Often when inoculating a BAP to observe hemoloysis patterns, investigators
will also stab several times through the agar using an inoculating loop.
This stab allows for the detection of streptolysin O, a specific hemolysin produced by Streptococcus pyogenes. This hemolysin is inactivated by O2 and is only seen subsurface (in an anaerobic
environment) around the stab mark. Note the oval-shaped areas of clearing
around the stab marks in the picture below; these are caused by streptolysin
O.
Bile
Esculin Agar
This is a medium that is both selective and differential. It tests
the ability of organisms to hydrolyze esculin in the presence
of bile. It is commonly used to identify members of the genus Enterococcus (E faecalis and E. faecium).
The first selective ingredient in this agar is bile, which inhibits
the growth of Gram-positives other than enterococci and some streptococci
species. The second selective ingredient is sodium azide. This
chemical inhibits the growth of Gram-negatives.
The differential ingredient is esculin. If an organism can hydrolyze
esculin in the presence of bile, the product esculetin is formed.
Esculetin reacts with ferric citrate (in the medium), forming
a phenolic iron complex which turns the entire slant dark brown
to black. The tube on the far right was inoculated with E.
faecalis (positive). The tube in the center was inoculated
with a bilie esculin negative organism and the tube on the left
was uninoculated.

TOP
Sulfur
Indole Motility Media (SIM)
This is a differential medium. It tests the ability of an organism
to do several things: reduce sulfur, produce indole and swim through
the agar (be motile). SIM is commonly used to differentiate members
of Enterobacteriaceae.
Sulfur can be reduced to H2S (hydrogen sulfide) either
by catabolism of the amino acid cysteine by the enzyme cysteine
desulfurase or by reduction of thiosulfate in anaerobic respiration.
If hydrogen sulfide is produced, a black color forms in the medium. Proteus mirabilis is positive for H2S production.
The organism pictured on the far left is positive for hydrogen
sulfide production.
Bacteria that have the enzyme tryptophanase, can convert the amino
acid, tryptophane to indole. Indole reacts with added Kovac’s
reagent to form rosindole dye which is red in color (indole +). Escherichia coli is indole positive. The organism pictured
second from left is E. coli and is indole positive.
SIM tubes are inoculated with a single stab to the bottom of the
tube. If an organism is motile than the growth will radiate from
the stab mark and make the entire tube appear turbid. Pseudomonas
aeruginosa and the strain of Proteus mirabilis that
we work with are motile.

TOP
Kliger’s Iron Agar (KIA)
This is a differential medium. It tests for organisms’ abilities
to ferment glucose and lactose to acid and acid plus gas end products.
It also allows for identification of sulfur reducers. This media is
commonly used to separate lactose fermenting members of the family Enterobacteriaceae
(e.g. Escherichia coli) from members that do not ferment lactose,
like Shigella dysenteriae. These lactose nonfermenting enterics
generally tend to be the more serious pathogens of the the gastrointestinal
tract.
The first differential ingredient, glucose, is in very short supply.
Organisms capable of fermenting this sugar will use it up within the
first few hours of incubation. Glucose fermentation will create acidic
byproducts that will turn the phenol red indicator in the media yelllow.
Thus, after the first few hours of incubation, the tube will be entirely
yellow. At this point, when the glucose has been all used up, the organism
must choose another food source. If the organism can ferment lactose,
this is the sugar it will choose. Lactose fermentation will continue
to produce acidic byproducts and the media will remain yellow (picture
on the far left below). If gas is produced as a result of glucose or
lactose fermentation, then fissures will appear in the agar or the agar
will be lifted off the bottom of the tube.
If an organism cannot use lactose as a food source it
will be forced to use the amino acids / proteins in the media. The deamination
of the amino acids creates NH3, a weak base, which causes
the medium to become alkaline. The alkaline pH causes the phenol red
indicator to begin to turn red. Since the incubation time is short (18-24
h), only the slant has a chance to turn red and not the entire tube.
Thus an organism that can ferment glucose but not lactose, will produce
a red slant and a yellow butt in a KIA tube (second from the left below).
These organisms are the more serious pathogens of the GIT such as Shigella
dysenteriae.
If an organism is capable of using neither glucose nor
lactose, the organism will use solely amino acids / proteins. The slant
of the tube will be red and the color of the butt will remain unchanged
(picture on the far right below). Pseudomonas aeruginosa is
an example of a nonfermenter.
KIA tubes are also capable of detecting the production
of H2S. It is seen as a black precipitate (second picture
from the right). Sometimes the black precipitate obscures the butt of
the tube. In such cases, the organisms should be considered positive
for glucose fermentation (yellow butt). Proteus mirabilis (pictured
here, second from right) is a glucose positive, lactose negative, sulfur
reducing enteric.

TOP
Nitrate
Broth
This is a differential medium. It is used to determine if an organism
is capable of reducing nitrate (NO3-) to
nitrite (NO2-) or other nitrogenous compounds
via the action of the enzyme nitratase (also called nitrate reductase).
This test is important in the identification of both Gram-positive
and Gram-negative species.
After incubation, these tubes are first inspected for the presence
of gas in the Durham tube. In the case of nonfermenters, this
is indicative of reduction of nitrate to nitrogen gas. However,
in many cases gas is produced by fermentation and further testing
is necessary to determine if reduction of nitrate has occurred.
This further testing includes the addition of sulfanilic acid
(often called nitrate I) and dimethyl-alpha-napthalamine (nitrate
II). If nitrite is present in the media, then it will react with
nitrate I and nitrate II to form a red compound. This is considered
a positive result. If no red color forms upon addition of nitrate
I and II, this indicates that either the NO3- has not been converted to NO2- (a negative
result), or that NO3- was converted to NO2- and then immediately reduced to some other, undetectable form
of nitrogen (also a positive result). In order to determine which
of the preceding is the case, elemental zinc is added to the broth.
Zinc will convert any remaining NO3- to
NO2- thus allowing nitrate I and nitrate
II to react with the NO2- and form the red
pigment (a verified negative result). If no color change occurs
upon addition of zinc then this means that the NO3- was converted to NO2- and then was converted
to some other undetectable form of nitrogen (a positive result).
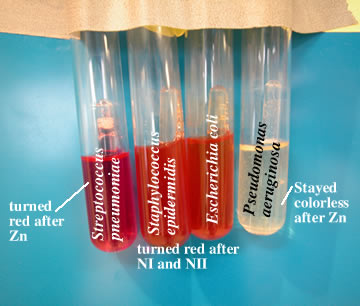
If the nitrate broth turns red (tubes pictured in the center)
after nitrate I and nitrate II are added, this color indicates
a positive result. If instead, the tube turns red (tube pictured
on the left) after the addition of Zn, this indicates a negative
result. If there is no color change in the tube after the addition
of nitrate I and nitrate II, the result is uncertain. If the tube
is colorless (picture on the right) after the addition of Zn this
indicates a positive test.
TOP
Catalase Test
This test is used to identify organisms that produce the enzyme, catalase.
This enzyme detoxifies hydrogen peroxide by breaking it down into water
and oxygen gas.

The bubbles resulting from production of oxygen gas clearly
indicate a catalase positive result. The sample on the right below is
catalase positive. The Staphylococcus spp. and the Micrococcus
spp. are catalase positive. The Streptococcus and
Enterococcus spp. are catalase negative.

TOP
Oxidase Test
This test is used to identify microorganisms containing the enzyme cytochrome
oxidase (important in the electron transport chain). It is commonly
used to distinguish between oxidase negative Enterobacteriaceae
and oxidase positive Pseudomadaceae.
Cytochrome oxidase transfers electrons from the electron transport chain
to oxygen (the final electron acceptor) and reduces it to water. In
the oxidase test, artificial electron donors and acceptors are provided.
When the electron donor is oxidized by cytochrome oxidase it turns a
dark purple. This is considered a positive result. In the picture below
the organism on the right (Pseudomonas aeruginosa) is oxidase
positive.

TOP
Coagulase test
Coagulase is an enzyme that clots blood plasma. This test is performed on Gram-positive, catalase positive
species to identify the coagulase positive Staphylococcus aureus. Coagulase is a virulence factor of S. aureus. The formation
of clot around an infection caused by this bacteria likely protects
it from phagocytosis. This test differentiates Staphylococcus aureus from other coagulase negative Staphylococcus species.
TOP
Taxos A (bacitracin sensitivity
testing)
This is a differential test used to distinguish between organisms sensitive
to the antibiotic bacitracin and those not. Bacitracin is a peptide
antibiotic produced by Bacillus subtilis. It inhibits cell wall
synthesis and disrupts the cell membrane. This test is commonly used
to distinguish between the b-hemolytic streptococci:
Streptococcus agalactiae (bacitracin resistant) and Streptococcus
pyogenes (bacitracin sensitive). The plate below was streaked with
Streptococcus pyogenes; notice the large zone of inhibition
surrounding the disk.
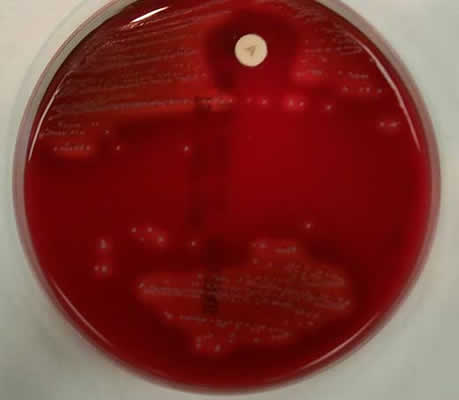
TOP
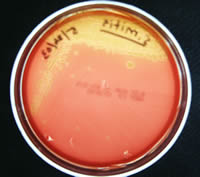
Taxos P (optochin sensitivity testing)
This is a differential test used to distinguish between organisms sensitive
to the antibiotic optochin and those not. This test is used to distinguish
Streptococcus pneumoniae (optochin sensitive (pictured on the right
below)) from other a-hemolytic streptococci
(optochin resistant (Streptococcus mitis is pictured on the left
below)).
TOP

MacConkey agar
This medium is both selective and differential. The selective
ingredients are the bile salts and the dye, crystal violet which
inhibit the growth of Gram-positive bacteria. The differential
ingredient is lactose. Fermentation of this sugar results in
an acidic pH and causes the pH indicator, neutral red,
to turn a bright pinky-red color. Thus organisms capable of
lactose fermentation such as Escherichia coli, form bright
pinky-red colonies (plate pictured on the left here). MacConkey
agar is commonly used to differentiate between the Enterobacteriaceae.
 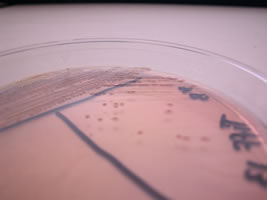
Organism on left is positive for lactose fermentation and that on the right is negative.
TOP
Simmon’s Citrate Agar
This is a defined medium used to determine if an organism can
use citrate as its sole carbon source. It is often used to differentiate
between members of Enterobacteriaceae. In organisms capable
of utilizing citrate as a carbon source, the enzyme citrase hydrolyzes
citrate into oxaoloacetic acid and acetic acid. The oxaloacetic
acid is then hydrolyzed into pyruvic acid and CO2.
If CO2 is produced, it reacts with components
of the medium to produce an alkaline compound (e.g. Na2CO3).
The alkaline pH turns the pH indicator (bromthymol blue) from
green to blue. This is a positive result (the tube on the right
is citrate positive). Klebsiella pneumoniae and Proteus
mirabilis are examples of citrate positive organisms. Escherichia coli and Shigella dysenteriae are citrate
negative.

TOP
Spirit Blue agar
This agar is used to identify organisms that are capable of producing
the enzyme lipase. This enzyme is secreted and hydrolyzes triglycerides
to glycerol and three long chain fatty acids. These compounds are
small enough to pass through the bacterial cell wall. Glycerol can
be converted into a glycolysis intermediate. The fatty acids can be
catabolized and their fragments can eventually enter the Kreb’s
cycle. Spirit blue agar contains an emulsion of olive oil and spirit
blue dye. Bacteria that produce lipase will hydrolyze the olive oil
and produce a halo around the bacterial growth. The Gram-positive
rod, Bacillus subtilis is lipase positive (pictured on the
right) The plate pictured on the left is lipase negative.
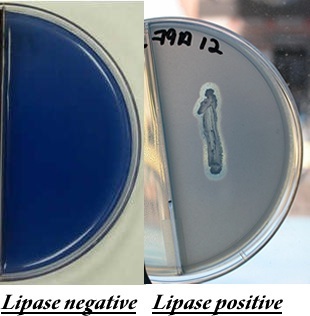
TOP
Starch hydrolysis test
This test is used to identify bacteria that can hydrolyze starch (amylose
and amylopectin) using the enzymes a-amylase
and oligo-1,6-glucosidase. Often used to differentiate species from
the genera Clostridium and Bacillus. Because of the
large size of amylose and amylopectin molecules, these organisms can
not pass through the bacterial cell wall. In order to use these starches
as a carbon source, bacteria must secrete a-amylase
and oligo-1,6-glucosidase into the extracellular space. These enzymes
break the starch molecules into smaller glucose subunits which can
then enter directly into the glycolytic pathway. In order to interpret
the results of the starch hydrolysis test, iodine must be added to
the agar. The iodine reacts with the starch to form a dark brown color.
Thus, hydrolysis of the starch will create a clear zone around the
bacterial growth. Bacillus subtilis is positive for starch
hydrolysis (pictured below on the left). The organism shown on the
right is negative for starch hydrolysis.
 
TOP
Methyl
Red / Voges-Proskauer (MR/VP)
This test is used to determine which fermentation pathway is used
to utilize glucose. In the mixed acid fermentation pathway, glucose
is fermented and produces several organic acids (lactic, acetic,
succinic, and formic acids). The stable production of enough acid
to overcome the phosphate buffer will result in a pH of below
4.4. If the pH indicator (methyl red) is added to an aliquot of
the culture broth and the pH is below 4.4, a red color will appear
(first picture, tube on the left). If the MR turns yellow, the
pH is above 6.0 and the mixed acid fermentation pathway has not
been utilized (first picture, tube on the right). The 2,3 butanediol
fermentation pathway will ferment glucose and produce a 2,3 butanediol
end product instead of organic acids. In order to test this pathway,
an aliquot of the MR/VP culture is removed and a-naphthol
and KOH are added. They are shaken together vigorously and set
aside for about one hour until the results can be read. The Voges-Proskauer
test detects the presence of acetoin, a precursor of 2,3 butanediol.
If the culture is positive for acetoin, it will turn “brownish-red
to pink” (tube on the left in the second picture). If the
culture is negative for acetoin, it will turn “brownish-green
to yellow” (tube on the left in the second picture). Note:
A culture will usually only be positive for one pathway:
either MR+ or VP+. Escherichia coli is MR+ and VP-. In
contrast, Enterobacter aerogenes and Klebsiella pneumoniae are MR- and VP+. Pseudomonas aeruginosa is a glucose
nonfermenter and is thus MR- and VP-.
 
TOP
CAMP Test
CAMP factor is a diffusible, heat-stable protein produced by
group B streptococci. This is a synergistic test between Staphylococcus
aureus and Streptococcus agalactiae. S. agalactiae produces CAMP factor. S. aureus produces sphingomyelin
C, which binds to red blood cell membranes. The two bacteria
are streaked at 90o angles of one another. They do
NOT touch. The CAMP factor produced by S. agalactiae enhances the beta-hemolysis of S. aureus by binding to
already damaged red blood cells. As a result, an arrow of beta-hemolysis
is produced between the two streaks. The test is presumptive
for S. agalactiae that produces CAMP factor.
In the picture here, Streptococcus agalactiae was
streaked throughout the top region of the plate and brought
down toward the center of the plate. Staphylococcus aureus was streaked in a straight line across the center of the plate.
Rings of hemolysis are evident all around S. aureus,
however the hemolysis if greatly enhanced (in an arrow shape)
where the S. agalactiae crosses the hemolysis rings.
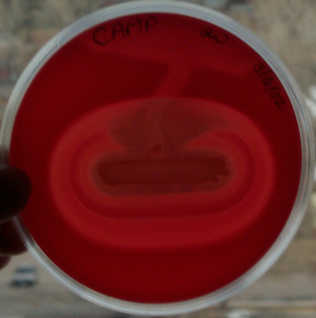
TOP
Urease
test
This test is used to identify bacteria capable of hydrolyzing
urea using the enzyme urease. It is commonly used to distinguish
the genus Proteus from other enteric bacteria. The hydrolysis
of urea forms the weak base, ammonia, as one of its products.
This weak base raises the pH of the media above 8.4 and the pH
indicator, phenol red, turns from yellow to pink. Proteus mirabilis is a rapid hydrolyzer of urea (center tube pictured here). The
tube on the far right was inoculated with a urease negative organism
and the tube on the far left was uninoculated.

TOP
Motility agar
is a differential
medium used to determine whether an organism is equipped with
flagella and thus capable of swimming away from a stab mark.
The results of motility agar are often difficult to interpret.
Generally, if the entire tube is turbid, this indicates that
the bacteria have moved away from the stab mark (are motile).
The organisms in the two tubes pictured on the right are motile.
If, however, the stab mark is clearly visible and the rest of
the tube is not turbid, the organism is likely nonmotile (tube
pictured on the left).

|

