Lecture 30 (14-Apr-13)
Required reading: McLaren and Peterson. 1994. Wolves, moose and tree rings on Isle Royale. Science 266: 1555-1558 (in WyoWeb folder)
Return to Main Index page Go back to notes for Lecture 29, 6 & 8-Apr Go forward to Lecture 31, 17-Apr-13
I will continue the section on population regulation by giving an overview of major concepts and by giving several interesting case histories. From the examples below it should become clear that a full understanding of regulation is very likely to require a community ecology point of view. The interactions among trophic levels (predator to herbivore prey, herbivore to plant resources) create a web of interconnected populations. Fully understanding the regulation of any one of them is likely to require considerable understanding of the many populations with which it interacts.
Some current theories of population regulation:
1) Potential regulating factors.
Concrete factors:
Nutrients and productivity
Environmental stress, disturbance and temporal heterogeneity
Space
Competition
Predation
Abstract factors:
Density-dependence
Density-independence
Top-down regulation (predator controlling herbivore controlling plants)
Bottom-up regulation (nutrients limiting plants limiting herbivores limiting predators)
2) Nutrient vs. disturbance models
Menge and Sutherland (1987)
focused on three major regulating forces --
disturbance (temporal heterogeneity), competition and predation. They argued
for a central role of environmental stress, with complexity inversely related
to environmental stress. For example, arctic, desert or alpine communities
should be simpler than temperate or tropical environments. They discuss
the implications of three levels of stress:
High stress: little effect of herbivores on plants, plants regulated
directly by environmental stress.
Moderate stress: herbivores relatively ineffective at regulating plants.
Plants will occur at high densities and be regulated largely by competition.
Low stress: Herbivores control plant populations and competition among plants should
be relatively rare.
Hairston, Smith and Slobodkin (1960) placed somewhat different emphases on the strengths of interactions among trophic levels. They felt that predators would regulate herbivores but that herbivores would have relatively little role in regulating plants, which would be regulated largely by competition for nutrients.
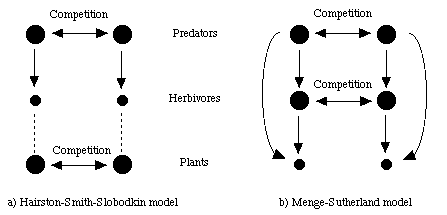
Fig. 30.1 indicates a schematic view of some of the differences between the two related models.
Fig. 30.1. Comparison and contrast between the Hairston model of regulation and the Menge-Sutherland (low stress environment) model. In the Hairston model, predators regulate herbivores (as indicated by downward solid arrows), but herbivores have relatively little effect on plant abundance (as indicated by dashed lines). In the Menge-Sutherland model, the top-down effect continues all the way to the effect of the herbivores on the plants. Notice also that the Menge-Sutherland model incorporates omnivory, as indicated by effects of predators on the plants. A grizzly-elk-plant chain might be a concrete example of the links indicated in the Menge-Sutherland diagram.Natural experiments by McQueen et al. (1989) examined the effect of a large winterkill of fish in Lake St. George, Ontario. Top predators such as bass, pike and yellow perch took five years to recover. During their relative absence planktivorous fish such as bluegills increased. Bluegills are primary carnivores (feeding on zooplankton), so they decreased the density of herbivorous zooplankton. That should, in turn, have led to an increase in phytoplankton, but the expected increase did not occur. Instead, the phytoplankton seemed to be limited by phosphorus levels. McQueen et al. therefore argued for both top down and bottom up effects. The removal of top predators cascaded down two trophic levels, but was dampened by bottom up regulation caused by nutrient limitations.
3) Top down regulation.
Fig. 30.2. Partial top-down regulation in a Canadian lake community. Panels (1 to 3 go top to bottom) show abundances of four categories of organisms:
1) piscivorous top predators such as bass, pike and yellow perch,
2) bluegills as primary carnivores feeding on zooplankton,
3) zooplankton, and 4) phytoplankton.
After a large die-off of fish in 1981 (second bar), the top predators took approximately five years to recover. As a result, the bluegills increased rapidly after the crash to a peak in 1984. The bluegill density in turn influenced the abundance of their prey, the zooplankton. The bottom of the food chain (phytoplankton) however, did not seem to respond clearly to the density of their predator (the zooplankton). Instead, phosphorus availability seemed to regulate the abundance of the phytoplankton. The bluegill-zooplankton-phytoplankton portion of this food web is closest to the Hairston model of Fig. 31.1a [After Krebs, 1994].
I will focus on several interesting examples of well-documented field studies of top-down regulation. Most examples of top down regulation involve a keystone predator. Note, however, that keystone species, and consequent top-down regulation may be relatively rare in many ecosystems. [See Mills et al. 1993 for an argument as to why focusing on keystone species may be a mistake in a conservation context].
Why the term "keystone"? In a Roman arch, the keystone is the essential support at the top of the arch. Weakness or removal of the keystone will cause the entire arch to collapse.
a) Snow geese on the arctic tundra. Kerbes et al. (1990) showed that huge increases in lesser snow geese caused by high overwintering survival has resulted in massive destruction of wetland systems along the shores of Hudson Bay in Canada. High overwinter survival, ironically, is due in part to successful management of wildlife refuges in the U.S., with supplemental feeding and consolidation and protection of habitat as major factors. This "top predator" in a very simple food chain (predator to vegetation) has had extensive impacts on many other species that depend on the wetlands.
b) Sea otters, urchins and kelp -- plus/minus orcas. Jim Estes and his colleagues have examined the role of sea otters, Enhydra lutris, as a keystone species in marine coastal environments. Sea urchins are a major food of the otters. The urchins in turn feed primarily on kelp. Following the development of the Russian fur trade from Alaska to California in the seventeenth century, otters were virtually eliminated from the Aleutian Islands in Alaska to the southern tip of their range in southern California. The effect on the coastline was dramatic. Kelp "forests" were virtually eliminated and the coastal ecosystem changed drastically (Duggins et al., 1989). The kelp forests provide a three-dimensional complex habitat and support a very large community of associates (just for example, herring depend on kelp as the spawning substrate -- no kelp, fewer herring). Recently an interesting higher level has been proposed. Orcas (killer whales) which had never really been present in Kachemak Bay, began preying on sea otters. Sea otters in Kachemak Bay have declined to 10% of their peak values in the late 80's. The prey switch by the orcas may represent a response to a crash of marine mammals (sea lions and fur seals). The importance of the impact of the orcas on the sea otters is controversial (sea otters have little fat and would at best represent a small snack for a killer whale). When I lived in Kachemak Bay (late '70's, very few otters) the waters were crystal clear and urchins were common. By the late 80's, the waters were cloudy green kelp forests (lots of otters). Now they're back to much clearer (few otters). Note that with four trophic levels it goes whales +, otters -, urchins +, kelp -. With three trophic levels the signs are reversed.
c) Wolves and moose. McLaren and Petersen (1994) studied the effect on wolves on moose and thereby on trees on Isle Royale in Lake Superior. They used a clever technique to show that wolves were a top down regulator. They looked at three paired sets of data: wolf abundance, moose abundance (both available from regular census sorts of data sets) and tree ring data. Over the course of three decades balsam fir ring growth suppression correlated with elevated moose densities. The ring suppression did not correlate with climate variables. Fir response lags varied across the island in a way that did correlate with east west differences in AET (Annual EvapoTranspiration; warmer early summer temperatures in the west, cooler boreal conditions at the east). Wolves in turn regulated the moose. Wolves were more common when older (>9 yrs) moose were available. In that case, calf survival was low and the fir trees were not suppressed. When the wolves depleted the older moose, calf survival rose again, and the moose population increased, followed by tree growth suppression. Cycles were: wolf maxima leading to moose minima leading to tree growth maxima, followed by wolf crash leading to moose maxima leading to tree growth minima. Finally, in a period when moose density was low, even in the absence of wolves, the tree growth DID correlate with climatic variation. This is essentially the "exception that proves the rule". Only when the higher links were removed did the system move from top down regulation to bottom up regulation.
References:
Duggins,
D.O., C.A. Simenstead, and J.A. Estes. 1989. Magnification of secondary production by kelp detritus in
coastal marine ecosystems. Science 245:
170-
Hairston, N.G., F.E. Smith, and L.B. Slobodkin. 1960. Community structure,
population control, and competition.
Am. Nat. 94: 421-425.
Kerbes, R.H., P.M. Kotanen, and R.L. Jeffries. 1990. Destruction of wetland habitats
by lesser snow geese: A keystone species on the west coast of Hudson Bay. J. Applied Ecol. 27: 242-258.
Krebs, C.J. 1994. Ecology (4th Edn.). Addison-Wesley, NY.
McLaren, B.E., and R.O. Peterson. 1994. Wolves, moose, and tree-rings on
Isle Royale. Science 266: 1555-1558.
McQueen, D.J., M.R.S. Johannes, J.R. Post, T.J. Stewart, and D.R.S. Lean. 1989.
Bottom-up and top-down impacts on freshwater
pelagic community structure. Ecol. Monogr. 59: 289-309.
Menge, B.A., and J.P. Sutherland. 1987. Community regulation: Variation in disturbance,
competition and predation in relation to environmental stress and recruitment. Am. Nat. 130: 730-757.
Mills, L.S., M.E. Soule, and D.F. Doak. 1993.
The keystone-species concept in ecology and conservation. Bioscience 43: 219-223.
§§§§ §§§§§§§§§§§ §§§§§§§§§§§§§ §§§§§§§§§§§§§§ §§§§§§§§§§§§§ §§§§§§§§
Return to top of page Go forward to Lecture 31, 17-Apr-13