Welcome to the Sun Lab
Information processing in the brain depends on inter-connected neuronal networks. The connections of neuronal networks are fine tuned during critical periods of brain development and maladaptively reorganized in neurological diseases. We want to understand how enviromental and genetic factors affect the refinement of neuronal connections and dynamically shape and mis-shape the the behaviour that these neuronal network subserve.
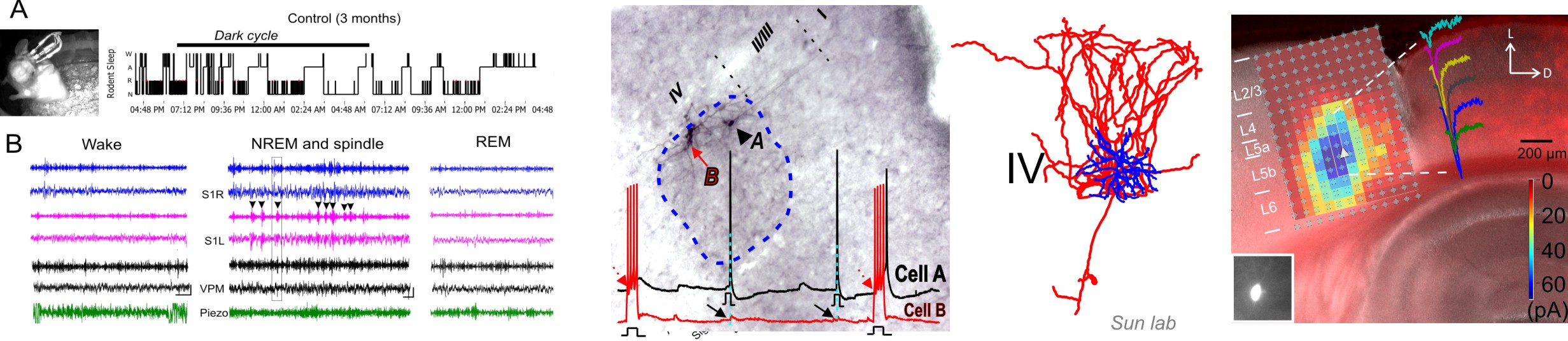
What we do
Even the simplest cognitive task requires communication between multiple sensory, motor, and association areas. Communication between these multiple areas is mediated by long-range projections acting on local neurons and is modulated by brain states. Our main research interest is to understand the adaptive changes in the properties of neural circuits at multiple spatial levels during the sensitive-periods of neurodevelopment and maladaptive changes associated with disease states. We have been using the rodent whisker-barrel somatosensory system to understand the organizing principles of cortical circuits and decipher the way they integrate into different circuits in the thalamocortical and sensorimotor cortex. We are interested in understanding how local and long-ranges cortical circuits are constructed, as well as how they adapt to sensory experiences and disease states. Our focus has been on GABA releasing inhibitory cells, because GABAergic synapses provide an inhibitory tone to local circuits, thereby determining the final output of local circuits. Distinct inhibitory neurons interact with excitatory neurons in space and time; they differentially contribute to the regulation of network oscillations and circuit dynamics6. Although the phenomena of functional differences in different interneurons have been measured across many brain regions, the mechanisms that underlie different activation patterns have been poorly understood. Abnormal inhibitory circuit wiring in cortical neurons are the key features of social and cognitive dysfunction such as epilepsy, autism and schizophrenia. Diverse genetic mutations cause different developmental brain disorders (DBD), yet many syndromes share similar intellectual and cognitive impairments. A common feature of these impairments involves disrupted vigilance and brain states. Thus, major efforts of our lab for the following decades will be to investigate how neural circuits, especially long-range circuits, undergo maladaptive changes in disease states across different states of consciousness.
To learn more about our research: Research
To know about the people: People and Life
**A new post-doc position is available (posted on May 15. 2017) see details HERE
