Grasshoppers of Wyoming and the West
Entomology
Greenstriped Grasshopper
Chortophaga viridifasciata (DeGeer)
 |
Distribution and Habitat
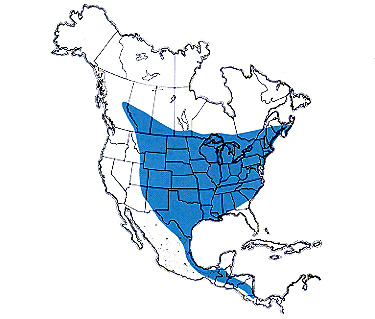 C. viridifasciata continental distribution map >
C. viridifasciata continental distribution map > The greenstriped grasshopper ranges widely in North America and extends southward into Central America. Inhabiting lands dominated by midgrasses, it is more prevalent in grass meadows and slopes of the eastern United States than in the extensive western prairies. Its distribution is patchy in the western region, as the species usually occupies small areas that receive additional moisture by overflow or by subirrigation. Patches of western wheatgrass growing in swales, around ponds, and along streams are preferred habitats of this grasshopper. It also inhabits stands of smooth brome under irrigation or located adjacent to irrigated crops and lawns.
Economic Importance
Because of its usually low numbers and patchy distribution, damage by the greenstriped grasshopper to rangeland forage is minimal and economically insignificant.In the eastern United States, where it is one of the most abundant and widespread species, it may cause some damage to pastures and hayfields. Records of its presence in fields of red clover and tobacco indicate it may become a minor pest of these crops. However, its food preferences suggest it may actually be feeding on weedy grasses that have invaded these crops. Of medium size, males of the greenstriped grasshopper collected in eastern Wyoming average 177 mg and females 607 mg (live weight).
Food Habits
The greenstriped grasshopper feeds mainly on grasses with a preference for succulent plants. Because of its wide distribution, it undoubtedly feeds on many different species of grass, but the only information available on host plants comes from a few localities.In southeastern North Dakota, where the species inhabits shortgrass areas of the sand prairie, it feeds chiefly on Kentucky bluegrass (44 percent). Additional host plants include foxtail barley, western wheatgrass, quackgrass, little bluestem, junegrass, needleleaf sedge, Penn sedge, and a forb, European sticktight. In the shortgrass prairie of northern Colorado, ten adults collected from a small swale dominated by western wheatgrass had crop contents of 91 percent western wheatgrass, 9 percent needleleaf sedge, and 0.2 percent blue grama. Direct observations of its feeding in Michigan revealed that the species fed upon orchardgrass and poverty oatgrass and a forb, peppermint. In northeastern Texas, food preference tests using several local plants showed that the first choice of greenstriped grasshoppers was new shoots of Johnsongrass. In the absence of Johnsongrass, the grasshoppers preferred a species of brome, Bromus catharticus. An unexpected result of the food selection tests was this grasshopper's voracious feeding on a forb, annual sowthistle, even in the presence of lush grasses.
The greenstriped grasshopper's method of attacking a grass is to climb the plant and begin feeding on the edge of a leaf about halfway up. A narrow leaf, like that of Kentucky bluegrass, is cut through, held by the front tarsi, and directed to the mouthparts. In the case of a broader leaf, such as a wheatgrass, the grasshopper may feed to the midrib and then continue to feed up the leaf to the tip. Occasionally, after consuming or dropping a cut section, the grasshopper will turn and feed head down on the attached portion. This grasshopper also feeds on ground litter, in which case a favorite item is a cut green leaf.
Dispersal and Migration
The greenstriped grasshopper possesses long wings that extend 2 to 8 mm beyond the end of the abdomen. A strong flier, it regularly disperses from its nymphal habitat to other areas. In Michigan the eggs hatch and the nymphs develop in moist swales and in the margins of marshes where Kentucky bluegrass grows luxuriantly. Numbers of the young adults fly out from these habitats into the uplands. Another example of dispersive flight is the observation on 8 May 1991 of a male sitting on a sidewalk of the University of Wyoming campus. This grasshopper had likely flown from a favorable habitat on the outskirts of Laramie.Adults take evasive flights in which the males fly distances of 10 to 30 feet and the females twice that far. The height of these flights ranges from 6 to 24 inches. Usually the grasshoppers crepitate in flight, but when quickly startled they fly without crepitation, which slows their speed. To the human ear the sound produced is a soft buzz. The insects dive and flutter to the ground in alighting. In eastern Wyoming an interesting observation was made of a male that rose up approximately 25 feet and then flew rapidly away from the habitat and beyond the observer's power of vision.
Males also fly and crepitate to attract the females. These flights are of short duration, no longer than two seconds, and most last only one second, and at heights up to 3 feet.
Identification
The greenstriped grasshopper is a green or brown, bandwinged species. The majority of females are green while the majority of males are brown (Fig. 6 and 7). Females are considerably larger than the males. The pronotal disk has posterior angle acute (Fig. 8); median carina is strong, equally elevated over its full length, and weakly cut once in front of middle; top of carina is usually light tan. Hind wings have basal area pale greenish yellow and the dark band is faded and incomplete (Fig. 9). Hind tibiae are bluish gray or red and have a tan annulus near proximal end.Nymphs (Fig. 1-5) are identifiable by their shape, structures, and color patterns.
1. Head. Face moderately slanting; antennae ensiform, ringed fuscous and white in instar I, fuscous ventrally, tan dorsally in instars II to V; compound eye with light spots and with light line that begins base of antenna and that usually divides eye into a light dorsal half and a dark ventral half.
2. Pronotum with disk tectate; median carina tan, moderately elevated and uncut; a tan band in green nymphs runs down middle of dorsum of thorax and abdomen; lateral carinae lacking in instars I to III, weak in instars IV and V.
3. Hind femur with outer medial area green, tan, or brown and with small dark spots.
4. General body color usually green or brown, sometimes tan (Fig. 2).
The early instars (I to III) of Chortophaga viridifasciata and Encoptolophus costalis appear similar; both are usually green and structurally similar. They may be separated by a few characteristics that differ. The antennae of E. costalis are clavate while those of C. viridifasciata are ensiform. The outer faces of the femur and tibia of the fore- and midlegs of E. costalis have four distinct longitudinal ridges, which usually have black lines between them. Those of C. viridifasciata have two distinct ridges, which are the upper and lower carina, and a third weak ridge between them with no black lines between the ridges. Instar I of E. costalis has the medial area of the hind femur pink in the distal half, and that of C. viridifasciata is entirely green. Instars IV and V are identifiable by the shape of the pronotum. The disk of E. costalis moderately, and that of C. viridifasciata is tectate (steep roof-like); the posterior angle of the disk in E. costalis is obtuse, and that of C. viridifasciata is acute.
Hatching
In eastern Wyoming, eggs of the greenstriped grasshopper hatch during the middle of July two weeks ahead of the velvetstriped grasshopper, Eritettix viridis, which frequently shares the same moist habitat. In southern states where two or more generations occur annually, hatching takes place earlier and at several times more through the growing season. In Texas, nymphs have been found throughout the year. Eggs deposited in southern states appear to develop without any diapause just as they do in the laboratory, where it is possible to obtain six generations in a year.
Nymphal Development
In the northern states the nymphs develop slowly, taking about 100 days to become fourth instars. In eastern Wyoming this stage is reached by the middle of October. Many of these nymphs molt to the fifth instar before cold weather compels them to enter dormancy. The nymphs appear to be very cold hardy. A fourth instar nymph collected close to the time of entering dormancy survived for 48 hours at 0°F. Nymphs become active in March and develop to the adult stage in early April. Laboratory research indicates that in winter these late instar nymphs may be in a facultative diapause, which is broken the following spring by an increasing photoperiod rather than the concomitant increasing temperature.Adults and Reproduction
Some adults may disperse from their nymphal habitats but others remain and reproduce there. Pair formation begins when females are attracted to crepitating males and approach them by flying and crepitating to signal a receptive response. When close, the pair begin to walk and hop toward each other. During courtship both males and females may also perform femur-tipping and other movements. The male stridulates as he approaches and when they touch, he mounts and they copulate. Copulations last 30 to 60 minutes.No information is available on how soon females begin oviposition or how many eggs they produce during their lifetime. In Indiana a female was observed to oviposit in damp sand at the margin of a pond. In the laboratory females readily oviposit in bare sandy loam soil. Females bore into the ground to a depth of one and one-fourth inches. Pods are one and one-eighth to one and one-fourth inches long and contain 25 eggs in the bottom half inch with the remainder of the pod being hardened froth. Eggs are light tan and 4 to 4.5 mm long (Fig. 10). In the northern part of its geographic range, this species has one generation annually and overwinters in the late nymphal stage.
Population Ecology
In the eastern United States the greenstriped grasshopper is one of the most abundant and widespread species of grasshoppers. Populations, however, do not grow to what pest managers call a grasshopper outbreak (25 adults or more per square yard). This may in part be due to a special auditory communication among the males that spaces individuals. The behavior may not only reduce competition among males for mates but may also limit population density.In western states populations are neither widespread nor abundant. The species is limited to small moist habitats in which densities of adults are less than one per square yard. As long as these habitats remain favorable, populations persist for years. Although many studies have addressed the behavior and physiological ecology of this species, apparently no special study has been made of its population ecology.
Daily Activity
Because of its wide distribution, the greenstriped grasshopper faces many different environmental hazards and extremes. In the north, e.g., eastern Colorado and Wyoming, late instar nymphs and adults must make adjustments to the cold and inclement weather of March and April. This they do by taking cover under litter, especially during the night. On April 4 in a smooth brome habitat next to an irrigated park lawn, grasshoppers were still hidden at 7 a.m. DST when soil surface temperature was 35°F and air temperature 1 inch high 33°F. By 8 a.m., when temperatures had risen (soil 45°F and air 40°F), the grasshoppers had emerged and were basking either horizontally on the ground or vertically, head up, on old culms of grass. They continued basking at least until 10 a.m. when soil temperature had risen to 67°F and air temperature 52°F During the middle of the day they feed and potter, spending most of this time on the ground.Further observations are needed to provide a complete picture of their daily activities. Because of low grasshopper numbers, pertinent facts will take considerable time to accumulate.
Selected References
Alexander, G. 1967. Cold hardiness in overwintering juvenile grasshoppers. Entomol. News 78: 147-154.Cantrall, I. J. 1943. The ecology of the Orthoptera and Dermaptera of the George Reserve, Michigan. Misc. Publ. Mus. Zool., Univ. Michigan, No. 54.
Carothers, E. E. 1923. Notes on the taxonomy, development and life history of certain Acrididae. Trans. Amer. Entomol. Soc. 49: 7-24.
Gangwere, S. K. 1961. A monograph on food selection in Orthoptera. Trans. Amer. Entomol. Soc. 87: 67-230.
Halliburton, W. H., and G. Alexander. 1964. Effect of photoperiod on molting of Chortophaga viridifasciata (DeGeer) (Orthoptera: Acrididae). Entomol. News 75: 133-137.
Isely, F. B. 1946. Differential feeding in relation to local distribution of grasshoppers. Ecology 27: 128-138.
Mulkern, G. B. 1980. Population fluctuations and competitive relationships of grasshopper species (Orthoptera: Acrididae). Transactions Amer. Entomol. Soc. 106: 1-41.
Mulkern, G. B., D. R. Toczek, M. A. Brusven. 1964. Biology and ecology of North Dakota grasshoppers. II. Food habits and preferences of grasshoppers associated with the sand hills prairie. North Dakota Agr. Exp. Stn. Research Report 11.
Otte, D. 1970. A comparative study of communicative behavior in grasshoppers. Misc. Publ. Mus. Zool., Univ. Michigan, No. 141.
Otte, D., and K. Williams. 1972. Environmentally induced color dimorphisms in grasshoppers. Syrbula admirabilis, Dichromorpha viridis, and Chortophaga viridifasciata. Ann. Entomol. Soc. Amer. 65: 1154-1161.
Steinberg, J. B., and R. B. Willey. 1974. Visual and acoustical social displays by the grasshopper Chortophaga viridifasciata (Acrididae: Oedipodinae). Canadian J. Zool. 52: 1145-1154.
Next Species in Subfamily: Derotmema haydeni

