Grasshoppers of Wyoming and the West
Entomology
Valley Grasshopper
Oedaleonotus enigma (Scudder)
 |
Link directly to photos of adults, nymphs, or eggs.
Distribution and Habitat
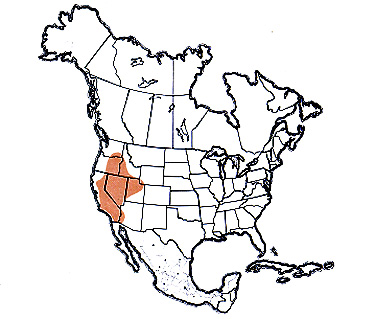 O. enigma continental distribution map >
O. enigma continental distribution map >
The valley grasshopper is a rangeland species, inhabiting the sagebrush-grass and other semiarid associations of the west. Native host plants include springparsley, balsamroot, big sagebrush, and rabbitbrush. The valley grasshopper has found several introduced weeds to its liking: redstem filaree, flixweed, and downy brome. The increase in number of favorable food plants appears to be an important factor in outbreaks of the species due to better nutrition. Abandoned farmland, Conservation Reserve Program (CRP) land, and foothills of the California coastal and Sierra Nevada ranges are especially favorable sites for development of large populations.
Economic Importance
High densities of the valley grasshopper on rangeland cause severe injury to forage plants. These grasshoppers are particularly damaging to young grasses and legumes in newly reseeded rangeland. The species has a high reproductive capacity. Density of adults may reach 20 per square yard and higher. Nymphs and adults in outbreak populations often migrate into alfalfa, cotton, grains, and vegetables, causing serious damage. The valley grasshopper may be beneficial at low densities as it prefers weeds for food, thereby thinning and reducing these strong competitors of valuable forage plants. No quantitative study of damage or benefits of this grasshopper, however, has been made.
The sizes attained by adults from one population to another are highly variable and are probably due to variations in environmental factors among habitats, such as temperature and quality and supply of food. Weights of adults collected in a drought-stricken habitat 12 miles south of Mountain Home, Idaho, and then caged and fed downy brome for 11 days averaged 365 mg for live males and 530 mg for live females (dry weight: males 110 mg, females 165 mg).
Food Habits
The valley grasshopper is primarily a forb and shrub feeder, but it also feeds to some extent on grasses. It has been observed to feed heavily on introduced weeds that often grow abundantly in its habitat. These include redstem filaree, tumble mustard, draba mustard, pepperweed, and downy brome. It likewise feeds heavily on lichen. In summer, when annual weeds have matured and dried, big sagebrush becomes an important host plant. In abandoned fields and CRP fields where big sagebrush no longer occurs, Russianthistle is often the only green plant available to the valley grasshopper in mid-summer. It does not feed upon this plant, but it does use it for roosting. Adult grasshoppers have been observed nibbling on the leaves of Russianthistle but never found to ingest any substantial amount of the plant. This may indicate that other members of the goosefoot family, including the sugar beet, are essentially immune to attack. Under drought conditions the grasshoppers resort to feeding on ground litter and dead or dying grasshoppers, and they will skirmish over an apple core thrown on the ground.
Direct observations and examination of crop contents have provided records of the valley grasshopper feeding on seven species of forbs, four shrubs, two grasses, one sedge, and one lichen. This list of food plants is undoubtedly incomplete.
To learn how the valley grasshopper attacks food plants, twigs of miniature rose (variety Meiponal) bearing leaves and blooms were transplanted 2 August 1991 in an abandoned field 12 miles south of Mountain Home, Idaho. Except for Russianthistle, the field contained only dry vegetation and ground litter. Shortly after being transplanted in the morning, the rose began to attract the adult grasshoppers. Some grasshoppers jumped onto the stem before making direct contact, while others kept crawling until making contact and then began to feed on lower leaves. In either case, individuals fed on the leaf edge beginning at the base or at the tip and often consumed the entire leaf before attacking another. A feeding grasshopper either stood on the ground or held onto the plant with the midlegs and hindlegs and used the front tarsi to hold the leaf and direct it to the mouthparts. Some grasshoppers cut through the petiole of leaves, which fell to the ground. Fallen leaves were eaten by grasshoppers still crawling on the ground. The grasshoppers also fed on the bracts and petals of flowers.
Valley grasshoppers were also offered transplanted spearmint. They fed on this plant in essentially the same way as on rose. They were observed walking to the mint and beginning to feed on the edge of the leaf down to mid rib and beyond. They also climbed the plant and began to feed. Grasshoppers on the rose and mint assumed various orientations suited to their feeding on the edges of leaves. At times they fed on the centers of leaves by folding them.
Dispersal and Migration
Slow dispersal of older nymphs and adults occurs almost daily. During an outbreak nymphs tend to migrate in concentrated bands of 20 to 30 per square yard. The grasshoppers may move in one direction while following or entering a draw with greener vegetation or they may spread out in all directions. Populations are known to move from abandoned fields and rangeland into irrigated crops.
Long-winged adults may disperse by flight. They are able to fly from deteriorating habitats into more favorable areas. Evidence for such flights comes from a drought-stricken habitat 12 miles south of Mountain Home, Idaho. On 24 June 1991 a dense population of young adults consisted of 54 percent macropterous and 46 percent brachypterous individuals. Sixteen days later the population had significantly decreased in density and consisted of only 18 percent macropterous and 82 percent brachypterous, indicating emigration out of the area by a majority of the long-winged adults.
Evasive flights of adults are straight, silent, and range from 4 to 8 feet in distance and 4 to 10 inches in height. On landing they face directly or diagonally away from the intruder. Brachypterous adults evade an intruder by jumping distances of 2 to 8 feet. The larger, stronger adults jump farther than the smaller ones.
In spite of its importance to integrated pest management of destructive populations, no special study of the dispersal and migration of the valley grasshopper has been made. We do not know the length nor height of flights, nor whether macropterous adults leave their original habitat individually, in groups, or en masse.
Identification
The adult valley grasshopper is a large, colorful, spurthroated grasshopper (Fig. 7 and 8). The tegmina range from short to long. Short tegmina are as long or longer than the pronotum. Seven other species of the genus Oedaleonotus can be distinguished by their possession of tegmina shorter than the pronotum, and the tegmina are usually narrow and widely separated. These seven species are distributed mainly in California.
Valley grasshoppers with long tegmina also have long hindwings that are functional organs of flight. Adults with short tegmina have even shorter, nonfunctional hindwings.
The anterior edge of the pronotum has a narrow, conspicuously white to cream-colored band, giving this grasshopper the appearance of wearing a clergyman's collar.
The medial area of the hind femur is marked with fuscous chevrons separated by light tan lines. The proximal end of the inner medial area and the lower marginal area are colored orange. The hind tibiae are blue. The cercus of the male is broad basally with apex abruptly narrowed and fingerlike (Fig. 9).
Nymphs are identifiable by their color patterns, structures, and shape (Fig. 1-6).
1. Head. Face slightly slanting, vertex and occiput with fuscous band down middle divided by narrow cream-colored line. Antennae filiform, first two segments pale tan or yellow with several darker spots, remainder of segments fuscous, each with narrow light ring on anterior edge. Compound eye with many cream-colored spots in brown reticulum, relatively large dark spot near center.
2. Thorax. Disk of pronotum dark brown with longitudinal cream-colored, narrow, fusiform band down middle. The entire dorsal light band begins on head and extends onto abdomen becoming faint posteriorly.
3. Hindleg. Medial area of hind femur with fuscous chevrons that are broken in middle at proximal half; hind tibia light gray with fuscous maculations.
4. General color light tan with fuscous spots and maculations. Shape is robust, pronotum widens posteriorly, matching wide meso- and metathorax.
Hatching
Overwintering as eggs in an advanced embryonic stage, valley grasshoppers hatch early in spring. Hatching may start in early April in California, Nevada, and Idaho and continue for a month or longer. Hatching usually occurs in the morning when air temperatures are between 45° and 90°F and soil temperatures are between 76° and 98°F.
Nymphal Development
Upon hatching in early spring the nymphs usually have a plentiful supply of food, but because of cool weather during this time of year they develop slowly, becoming adults in about 42 to 50 days. Compared with later-hatching species, the valley grasshopper has a long nymphal period, due to both the cooler temperatures and to the greater number of nymphal instars, six instead of the usual five. The proportion of males to females is nearly 1:1. In different years, the proportion of short-winged adults to long-winged adults ranges from all short-winged to over 50 percent long-winged. Laboratory tests indicate that temperature may be one factor that influences this proportion. Cooler developmental temperatures (constant 80°F) result in greater proportions of long-winged adults, while warmer temperatures (constant 100°F) result in more short-winged adults.
Adults and Reproduction
Although many adult valley grasshoppers disperse from their nymphal habitats, variable numbers remain and reproduce, even in a deteriorating habitat. In a north-central Nevada site in 1954 when average temperatures were slightly above normal, the first adults appeared on 22 May, mating pairs were noted 20 days later, and egg deposition began two weeks after mating. Eggs begin development upon being laid in summer; by fall they reach an advanced developmental stage (after blastokinesis, stage 23), and then go into diapause. During winter the diapause is broken and warming weather in spring enables the eggs to complete embryonic development.
Females oviposit in bare ground adjacent to the base of shrubs and weeds, under cover of low growing forbs, such as turkey mullein, and around and under rocks. The pods, placed horizontally one-eighth to one-quarter inch below the soil surface, contain 16 to 22 eggs each. Anterior ends of the eggs face diagonally toward the soil surface. On hatching, the nymphs emerge from the side of the pod rather than through the end as is usually the case with other species of grasshoppers. The pods are slightly curved, short, and wide - one-half to five-eighths inch long and one-eighth to three-sixteenths inch diameter. Eggs are olive tan and 4.8 to 5.2 mm long (Fig. 10).
Population Ecology
The valley grasshopper has the capacity to increase to high numbers on rangeland and cause serious outbreaks. Densities may rise to over 20 adults per square yard. Outbreaks may be terminated by drought. At these times vegetation turns brown and desiccates, causing nutritional problems for the grasshoppers. Older instars and adults may disperse to survive in other areas with green host plants. Predation may also effectively end an outbreak. During June of 1954 three species of digger wasps (Tachysphex), which provision their offspring with nymphs of the valley grasshopper, reduced a population in south central Idaho from 25 per square yard to 3 per square yard. Large numbers of wasps emerged in June 1955 but they had virtually no grasshoppers on which to prey. Field studies indicate that the principal dipterous parasite is the tangleveined fly, Neorhynchocephalus sackenii (Williston); rate of parasitism during four years of study was variable, ranging from 0 to 24 percent.
Regrettably, no population has been studied for more than three years, a period too brief to gain useful information and insights into the population ecology of this grasshopper. Important questions for integrated management remain unanswered - how long outbreaks last, how long populations remain at low densities, and how many years are required for populations to grow from low to high numbers.
Daily Activity
The behavior of adults inhabiting a drought-stricken abandoned field 12 miles south of Mountain Home, Idaho was observed for three days in August 1991. Early food plants, tumble mustard and downy brome, had matured and dried and only Russianthistle remained green. Valley grasshoppers refused to eat this plant but regularly used it for roosting.
The majority of valley grasshoppers spent the night roosting head-up on the main or secondary stems of Russianthistle plants, but a small number rested on the ground exposed or under a canopy of Russianthistle. At daybreak, before the sun had risen, the adults were quietly resting in different orientations. At this time (6 a.m. DST), surface soil temperatures ranged from 52° to 64°F and air temperatures 1 inch from the ground ranged from 52° to 62°F. An hour later the vertically roosting grasshoppers assumed a basking orientation by adjusting their position so that the sun's rays shone perpendicularly on their sides. They remained quietly basking on the Russianthistle plants for an hour, then began to climb down to the ground, head first, where they again basked by turning a side perpendicularly to the sun's rays and lowering the exposed hindleg to the ground. A few spread this hindleg to the side, exposing even more of the abdomen.
Regular activities of pottering, feeding, and mating began at 8:30 a.m. when surface soil temperatures had risen to 82°F and air temperature 1 inch above ground level to 67°F. The grasshoppers fed on ground litter and on dead or dying bigheaded grasshoppers, Aulocara elliotti. By 10:45 a.m. ground temperatures rose to 110°F, inducing grasshoppers to elevate their bodies off the soil surface by stilting (raising up on their legs). By noon the soil surface temperature was 130°F, and the majority of the grasshoppers had climbed or jumped into Russianthistle, resting at heights of 4 to 7 inches. A few crawled into the shade of Russianthistle and remained on the ground. When temperatures moderated in late afternoon the grasshoppers again became active pottering, feeding, and mating. By 8 p.m., an hour before sunset, the majority of grasshoppers were roosting on Russianthistle and remained in these positions for the night.
Selected References
Brusven, M. A. 1972. Differentiation and ecology of common Catantopinae and Cyrtacanthacridinae nymphs (Orthoptera: Acrididae) of Idaho and adjacent areas. Melanderia 9: 1-31.
Brusven, M. A. and J. D. Lamley. 1971. The food habits and ecology of grasshoppers from southern Idaho rangeland. Univ. Idaho in cooperation with USDA, Project Completion Report 1967-1971.
Hostetter, D. L., S. L. Breeding, J. A. Onsager, D. K. Broemeling, and S. L. Zugoni. 1990. Impact of insect parasites and predators on grasshopper populations. I (Idaho). 1990 Annual Report Cooperative Grasshopper Integrated Pest Management Project, pp. 265-273.
Middlekauff, W.W. 1958. Biology and ecology of several species of California rangeland grasshoppers. Pan-Pacific Entomol. 34: 1-11.
Newton, R. C. 1956. Digger wasps, Tachysphex spp., as predators of a range grasshopper in Idaho. J. Econ. Entomol. 49: 615-619.
Seaton, Lee. 1955. Life history and habits of Oedaleonotus enigma. USDA ARS Special Report Z-33.
Sheldon, J. K. and L. E. Rogers. 1978. Grasshopper food habits within a shrub-steppe community. Oecologia 32: 85-92.
Xiangchu, Y. and R. L. Smith. 1989. Three new grasshopper species from the Western United States (Orthoptera: Acrididae). Pan-Pacific Entomologist 65: 166-171.
Next Species in Subfamily: Phoetaliotes nebrascensis

